Hidradenitis Suppurativa (HS): Can sonelokimab/SLK raise the bar on treatment efficacy?
- Jul 23, 2025
- 6 min read
Key takeaways
HS is a common, chronic skin condition that causes painful lumps/abscess under the skin.
Existing biologics treatments are not curative, but provide symptoms control. For moderate-to-severe patients, 1L treatment is Humira (or its biosimilars) and 2L treatment is IL-17 inhibitors including Cosentyx and Bimzelx.
SLK (sonelokimab) is a nanobody IL-17A/F inhibitor being developed by Moonlake Therapeutics. It is being evaluated in Ph-3 trials in the hope of demonstrating greater efficacy than existing treatments.
I expect SLK Ph-3 to achieve 40-45% unadjusted HiSCR75*, demonstrating greater efficacy than other IL-17 inhibitors but similar efficacy as Humira.
SLK Ph-2 already showed ~45% unadjusted HiSCR75*, which is +5-20 p.p. higher compared to other IL-17 inhibitors, and +5-10 p.p. higher compared to Humira.
SLK Ph-3 may show similar or slightly lower efficacy as its Ph-2 results. Ph-3 may show 5 p.p. higher HiSCR75 due to a longer treatment duration (16 vs. 12 weeks), but this could be offset by a larger N size and lower baseline antibiotic use.
SLK Ph-3 trial protocol changes may further impact efficacy, but impact direction is unclear. Protocol details were not disclosed and MLTX only stated that it is similar to the Ph-2 protocol.
* HiSCR75 = % patients with >75% decrease in abscess and inflammatory nodule #, with no # increase in abscesses or draining fistulae.
HS is a common, chronic skin condition that causes painful lumps/abscess under the skin (source)
HS affects 1-2% of people in the US, onsetting during ages 16-40 and affecting more women and minority ethnicities .
HS significantly impacts quality of life in terms of physical movement limitations and psychological depression. The condition usually affects skin areas where the skin rubs together (e.g., armpits, groin, buttocks, breasts). The condition starts to develop with hair follicle blockage, followed by lumps, abscess, and tunnels under the skin. The lumps cause great pain (e.g., much worse than that of psoriasis), odor, and swelling.
The cause of HS is unclear, but likely connected to genetic predisposition, hormonal changes, smoking history, or overweight. HS patients often have comorbidities in obesity, PCOS, depression, and anxiety.
HS is underdiagnosed and existing treatments can relieve some symptoms, as measured by HiSCR50/75/90
HS diagnosis is often delayed by years due to low disease awareness, misdiagnosis (e.g., no lab test to diagnose HS), and low access to dermatologists.
Existing HS treatments alleviate some symptoms and are not curative
Given that the pathogenic cause is not understood, HS has been treated as an inflammatory disease and existing therapies aim to alleviate symptoms as opposed to curing the disease.
For early-stage disease, topical antibiotic is the first-line treatment.
Efficacy is usually measured by HiSCR50, HiSCR75, or HiSCR90. It calculates the % patients with >50%, 75% or 90% decrease in abscess and inflammatory nodule #, with no # increase in abscesses or draining tunnels. These metrics are typically used as primary endpoints in clinical trials.
Other efficacy measures include the DLQI (a questionnaire assessing quality of life), and change in HSSI4 (a composite score based on the number of inflammatory nodules, abscesses, and draining tunnels).
For moderate or severe stages, 1L treatment is Humira/biosimilars, and 2L treatment is IL-17 inhibitors (Cosentyx, Bimzelx)
TNF-α inhibitor (Humira and its biosimilars) is 1L, given its extensive real-world experience since 2015 approval and lower cost.
IL-17 inhibitors (Consentyx and Bimzelx) are newer drugs approved in 2023 and 2024. They are gaining market share, as 30-60% of HS patients fail Humira and have no alternative treatment option.
JAK1 inhibitor is a potential 3rd drug class
IL-1α / β inhibitor is a potential 4th drug class


Sonelokimab (SLK) by Moonlake Therapeutics is the latest pipeline asset trying to raise the bar for symptom control in HS.
SLK belongs to the IL-17 inhibitor drug class, along with Cosentyx (IL-17A inhibitor) and Bimzelx (IL-17 A/F dual inhibitor). However, its drug design is unique in that:
Compared to Cosentyx, SLK is an IL-17 A/F dual inhibitor, which could inhibit all IL-17 dimers. Moonlake Therapeutics believes that a broader coverage offers more complete IL-17 pathway inhibition.
SLK and Bimzelx are both dual inhibitors, but SLK utilize nanobody as opposed to a traditional antibody to bind to IL-17 targets. Moonlake Therapeutics believes that having a physically smaller antibody can achieve deeper tissue penetration and thereby greater efficacy.
SLK is being evaluated in Ph-3 studies to support a potential FDA approval.
Trial evaluates SLK against placebo as control arm
Primary endpoint is HiSCR75 at 16 weeks
SLK Ph-2 showed +5-20 p.p. higher HiSCR75 compared to other IL-17 inhibitors (Cosentyx and Bimzelx), and +5-10 p.p. higher HiSCR75 compared to Humira.
SLK vs Cosentyx: SLK Ph-2 showed +20 p.p. higher HiSCR50 and +20 p.p. higher HiSCR75
SLK Ph-2 vs Cosentyx Ph-1/2: Given prior approval in other immunology indications, Cosentyx only ran a Ph-1/2 trial for HS with 20 patients. Cosentyx Ph-2 efficacy was similar to SLK Ph-2, but likely overstated due to trial design -- e.g., an open-label study and LOCF statistical analysis method (i.e., if a patient drops out, their last recorded outcome is carried forward to all subsequent time points).
SLK Ph-2 vs. Cosentyx Ph-3: At each HiSCR level, SLK achieved +20 p.p. greater clinical response rates for non-adjusted and placebo-adj results.
Furthermore, SLK efficacy might have been understated due to a shorter treatment period (12 weeks vs 16 weeks), the use of NRI statistical method (i.e., consider all patients with missing data as non-responders), and not requiring all patients to use antiseptics during trial.



SLK vs Bimzelx: SLK Ph-2 showed +10-15 p.p. higher HiSCR50 and +5-10 p.p. higher HiSCR75
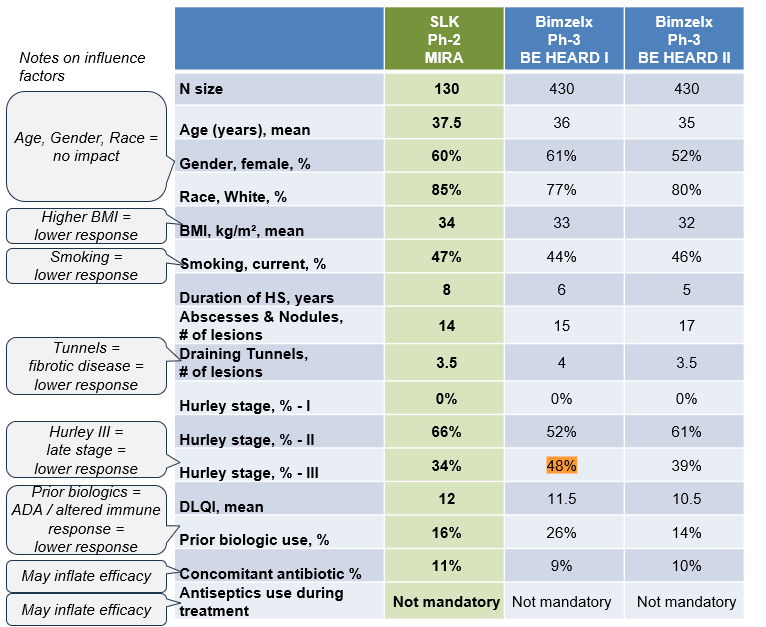
SLK Ph-2 vs. Bimzelx Ph-2: At first glance, SLK appeared less effective than Bimzelx across HiSCR50, HiSCR75, and HiSCR90. However, Bimzelx's efficacy was inflated due to trial design decisions -- e.g., using per-protocol patients vs. ITT for data analysis, using Bayesian augmentation to incorporate external placebo data (from Humira Ph-3 trials), and mandating antiseptics usage during trial.
SLK Ph-2 vs. Bimzelx Ph-3: SLK showed +10-15 p.p. more in HiSCR50 and +5-10 p.p. more in HiSCR75 than Bimzelx. Furthermore, SLK efficacy might have been understated due to a shorter treatment period (12 weeks vs. 16 weeks) and the NRI statistical method (i.e., consider all patients with missing data as non-responders).




SLK vs. Humira: SLK Ph-2 showed +7 p.p. higher HiSCR50 and +8 p.p. higher HiSCR75
SLK treatment arm vs Humira control arm in Ph-2 result: In SLK's Ph-2 MIRA trial, Humira was included as a control arm. SLK arm showed +8 p.p. more in HiSCR50 and +7p.p. more in HiSCR75. Patient baseline details were not disclosed for the Humira control arm, so it's unclear if patient characteristics had impacted the results.
SLK Ph-2 vs Humira Ph-3 result: In cross-trial comparison between SLK Ph-2 MIRA trial and Humira Ph-3 trials, SLK showed +7 p.p. more in HiSCR50 and +8 p.p. more in HiSCR75.
Humira had two Phase 3 trials: PIONEER I and PIONEER II. HCPs commonly benchmark Humira’s efficacy against PIONEER II, as patients in PIONEER I were sicker and did not receive concomitant antibiotics at baseline, making them potentially less responsive to treatment.



I expect SLK Ph-3 to hold similar or slightly lower efficacy than its Ph-2 results -- e.g., 0-15 p.p. higher HiSCR75 compared to other IL-17 inhibitors, and 0-5 p.p. higher HiSCR75 compared to Humira.
SLK Ph-3 HiSCR75 may increase by 5 p.p. by extending treatment duration from 12 weeks to 16 weeks. In SLK Ph-2, HiSCR75 (unadjusted) increased from 43% in week 12 to 57% in week 24, suggesting an average of +5 p.p. for every 4 additional weeks.
SLK Phase 3 HiSCR75 rates may decrease by 2–3 p.p. due to lower concomitant antibiotic use (8% in Phase 3 vs. 12% in Phase 2). In Humira’s Phase 3 trials, HiSCR75 was 25% in PIONEER I (0% antibiotic use) and 35% in PIONEER II (20% antibiotic use), suggesting an average change of 2–3 p.p. for every 5% difference in concomitant antibiotic usage. Other patient baseline characteristics were similar between the trials.
SLK Phase 3 HiSCR75 rates may decrease by a few p.p. due to larger sample size (520 Phase 3 vs. 60 in Phase 2). Efficacy often declines in larger trials due to greater variability in patient characteristics and protocol execution variability across more trial sites.
SLK Ph-3 trial protocol's impact on efficacy is unclear. Protocol details were not disclosed and MLTX only stated that it is similar to the Ph-2 protocol.


Comments