Myotonic Dystrophy Type 1 (DM1): DYNE-101 vs. AOC-1001
- Jul 10, 2025
- 7 min read
Updated: Jul 10, 2025
Main takeaways
DM1 is a rare muscular disorder that leads to muscle weakness, myotonia (difficulty relaxing muscles), and mobility issues.
DYNE-101 and AOC-1001 are the most advanced assets in clinical development. Both are antibody-oligonucleotide conjugates (AOC) that deploy a novel TfR1-targeting mechanism, but use different payload technologies (ASO vs. siRNA)
Both assets have completed Ph-1/2 trials and are being evaluated in pivotal trials.
In Ph-1/2 trials with small n sizes, DYNE-101 and AOC-1001 demonstrated similar and moderate clinical efficacy. Safety profiles were similar, with potential SAEs linked to the TfR1-targeting mechanism.
In pivotal trials, I expect DYNE-101 and AOC-1001 to show statistically significant and similar efficacy results, but AOC-1001 may carry higher safety risk than DYNE-101.
DM1 is a rare muscular disorder that causes significant challenges related to muscle weakness, myotonia, and immobility.
DM1 affects 120K patients in the U.S. and Europe.
DM1 is a genetic disease associated with DMPK gene mutation. In DM1 patients, The DNA’s DMPK gene is mutated and leads to abnormally long mRNAs. These mutant mRNAs accumulate in the nucleus, trap splicing proteins, and prevent them from splicing other mRNAs. Without splicing, other mRNAs are mis-spliced and produce dysfunctional proteins in the body. (note: splicing is part of the process of making functional mRNAs)
While DM1 does not affect life expectancy, it significantly reduces quality of life. The most common clinical symptoms include muscle weakness, myotonia (difficulty relaxing muscles), fatigue, and balance issues. In everyday life, patients struggle with opening jars or turning knobs, climbing stairs, and standing for extended periods. [source]. In addition, DM1 patients may suffer from cardiac muscle disorders (e.g., arrhythmia), smooth muscle disorders (e.g., pulmonary disease, gastrointestinal disease), and neurological dysfunctions.
DYNE-101 and AOC-1001 are the most advanced assets in development
As of 2025, DM1 has no approved therapy.
Clinical development has focused on two mechanisms of action, forming a pipeline of six assets. AOC-1001 from Avidity Biosciences and DYNE-101 from Dyne Therapeutics are leading the pipeline and represent the first-in-class assets for their respective mechanisms.

DYNE-101 and AOC-1001 are antibody-oligonucleotide conjugates (AOC) that deploy a novel TfR1-targeting mechanism, but use different payload technologies.
Both assets are designed for delivery into skeletal, cardiac, and smooth muscle cells. DYNE-101 may also deliver into the brain / CNS.
Both DYNE-101 and AOC-1001 target TfR1 receptors to enter cells. TfR1-targeting is a novel mechanism, with no FDA-approved drugs or other Ph-3/pivotal trial using this approach as of 2025.
However, DYNE-101 uses ASO payload (a small synthetic DNA strand) to degrade DMPK mRNA directly in the cell nucleus. ASO technology has been validated in neuromuscular disorders with six FDA-approved drugs.
Meanwhile, AOC-1001 uses siRNA payload (small RNA segments) to degrade DMPK mRNA in the cytoplasm, which also indirectly reduces mRNA production in the nucleus. While FDA has approved siRNA drugs, the technology has not been approved for neuromuscular disorders.


main sources:
DYNE-101 and AOC-1001 are being evaluated in pivotal clinical trials
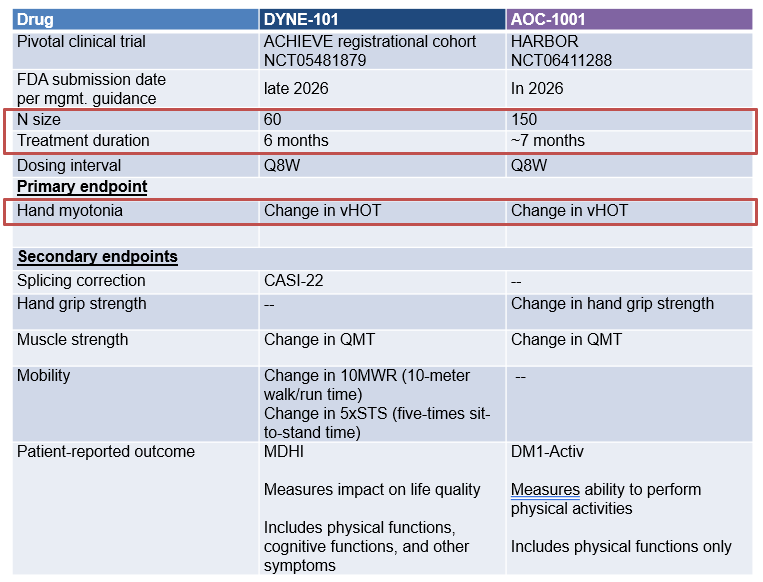
AOC-1001 is being evaluated in a full Ph-3 trial with a larger dataset, while DYNE-101 is in a Ph-1/2 expansion cohort with a smaller dataset (for accelerated approval).
Both assets will be evaluated against placebo over 6-7 months of treatment
Change in vHOT is the primary endpoint. vHOT is the time to open a fist, a standard assessment of hand myotonia. This endpoint is clinically relevant and serves as an early indicator of treatment efficacy (as it tends to respond quickly).
Change in QMT (quantitative muscle testing) is a common secondary endpoint. In addition, DYNE-101 will measure PRO in physical and cognitive functions, and change in mobility. AOC-1001 will measure PRO in physical functions and change in hand grip strength.
(PRO = patient-reported outcome)
Efficacy assessment:
In pivotal trials, I expect DYNE-101 and AOC-1001 to show similar efficacy, with significant improvement in vHOT and numerical improvements in secondary endpoints. However, efficacy will be moderate and unlikely curative.
Both assets have already been evaluated in Ph-1/2 trials over 6 months.
In Ph-1/2, DYNE-101 and AOC-1001 were evaluated against placebo for 6 months. However, N sizes were small and there was no formal statistical testing during the study.

At baseline, DYNE-101 patients had milder disease symptoms compared to AOC-1001 patients. Healthier patients in DYNE-101 trial may respond better to treatment and support greater efficacy results.

In Phase 1/2 trials, DYNE-101 and AOC-1001 showed significant improvement in vHOT, and numerical improvements in QMT and PRO.
For vHOT, both assets showed a 30-40% improvement by 6 months, indicating myotonia correction. In post-hoc analyses, the vHOT improvements were statistically significant across time points.

For QMT, both assets showed a ~10% improvement by 6 months, indicating a modest gain in muscle strength. The improvements were not statistically significant across time points (i.e., the treatment arm and placebo arm had overlapping standard error bars). However, the curve trends were durable, with continued improvement and growing separation from placebo.

For PRO, both assets saw patients report daily functioning improvements, but results were not comparable due to different methodologies (MDHI for DYNE-101 vs. DM1-Activ for AOC-1001). In particular, DYNE-101 tracked cognitive benefits, but the effect was not sustained over time.


Neither asset showed improvement for 10mWRT over 6 months, suggesting no impact on mobility.

In Phase 1/2 trials, efficacy improvements were similar between DYNE-101 and AOC-1001
Across the common clinical endpoints, DYNE-101 showed numerically greater improvements compared to AOC-1001. However, these differences were likely NOT significant due to wide error intervals from small sample sizes. Furthermore, it remained unclear whether DYNE-101's higher numerical values reflected a true efficacy advantage or the influence of a healthier patient population in the trial.

In Phase 1/2 trials, clinical improvements were moderate in both assets, which could be explained by partial corrections at the molecular level
Both assets achieved a 20–30% reduction in abnormal DMPK mRNA levels.
Both also showed a 30–40% reduction in mRNA mis-splicing activities, but the values were not directly comparable due to different measurement methods (CASI-22 vs. splice score).
These results suggested that the mechanism of actions were working, but impact was partial and lower than the theoretical goal of near-complete (≈100%) correction.

In pivotal trials, I expect DYNE-101 and AOC-1001 to show similar efficacy, with significant improvement in vHOT and numerical improvements in secondary endpoints.
Ph-1/2 trials have demonstrated significant vHOT improvement over 6 months, along with numerical improvements in some secondary endpoints
For the primary endpoint (vHOT), the significant improvement over multiple time points supports the likelihood of replicating efficacy in larger trials.
For secondary endpoints QMT and PRO, the durability of curve trends (e.g., continued improvement, continued curve separation from placebo) supports the likelihood of seeing numerical improvements in larger trials
Other secondary endpoints in mobility, cognitive function, and hand grip strength may not contribute meaningfully to efficacy profile. DYNE-101 is unlikely to show improvement in mobility and cognitive function. AOC-101 might show improvement in hand grip strength, but the endpoint is unlikely to add differentiation on its own.
The pivotal trial designs closely mirror those of the Phase 1/2 studies.
DYNE-101 is maintaining the same design, including patient eligibility criteria, treatment schedule, and dosing frequency.
AOC-1001 will increase the dosing frequency from Q13W in Phase 1/2 to Q8W in the pivotal trial. This adjustment might help sustain treatment activity and enhance efficacy, but there is no pre-existing evidence for support.
main sources:
Safety assessment:
I expect DYNE-101 and AOC-1001 to carry major safety risks, with AOC-1001 posing a higher risk in pivotal trial.
In Ph-1/2, DYNE-101 and AOC-1001 demonstrated similar safety profiles, with major safety risks potentially linked to TfR1-targeting mechanism.
Both assets presented similar overall AE rates
Neither asset resulted in patient death or high treatment discontinuation rate (<10%)
AOC-1001 had a lower SAE rate than DYNE-101 (5% vs 9%), but the small trial size (n=20 vs 55) might have limited the detection of SAEs.
Both assets showed treatment-related AE rate of 60%, and presented similar AE types (e.g., biopsy pain, infusion site reactions, influenza/cold, anemia, headache).
Both assets saw serious safety risk that might be linked to TfR1-targeting mechanism
TfR1 is widely expressed on cells and regulates cellular iron uptake (source). Targeting TfR1 may disrupt iron balance, leading to iron deficiency or overload.
Iron deficiency can cause anemia, headache and fatigue (source), which were AEs observed in both trials. AOC-1001 showed a higher anemia rate than DYNE-101 (30% vs <20%).
Severe or untreated iron deficiency can cause arrhythmia, heart failure, and serious infections (source). Evidently, DYNE-101 reported SAEs including atrioventricular block (a type of arrhythmia), pneumonia, and influenza. While the pulmonary embolism SAE may not be directly linked to iron deficiency, arrhythmia is a known risk factor (source). AOC-1001 reported one SAE involving brain stroke, and anemia is a known risk factor for stroke.
DYNE-101 showed slightly higher rates of inflammation, infection, and liver toxicity compared to AOC-1001, which might be linked to their ASO and siRNA payloads (source, chatgpt).
Injection site reaction is a common risk for ASO and siRNA. It was a top AE in both trials.
Immune-related effects is common for ASO, and DYNE-101 showed higher rates of cold and influenza compared to AOC-1001.
Liver and kidney toxicity are known risks for ASO and siRNA therapies. In both trials, a subset of patients experienced elevated liver enzymes. DYNE-101 also reported a SAE of hyponatremia (low sodium), which may suggest impaired fluid and electrolyte regulation due to liver or kidney stress.

In pivotal trials, I expect higher safety risk in AOC-1001 than DYNE-101
Since many AEs might be tied to drug MoA, AE rates are likely to persist in pivotal trials. Neither asset has disclosed plans to monitor iron imbalance directly, increasing the risk of unaddressed complications. While AOC-1001 is introducing more frequent safety check-ins and routine MRI monitoring in response to a prior SAE, the specifics of these measures remain unclear.
In addition, AOC-1001 introduces four factors that could elevate SAE risk in its pivotal study:
Higher anemia rates: In Ph/1-2, AOC-1001 showed a 30% anemia rate vs. <20% for DYNE-101, which could result in more SAEs in a larger population. Although AOC-1001 had a lower SAE rate in Phase 1/2, the small sample size may have masked SAEs.
Looser cardiovascular exclusion criteria: AOC-1001 previously excluded patients with uncontrolled hypertension but now excludes only those with heart failure history. This change may allow enrollment of patients with non-HF cardiac conditions who are still vulnerable to iron-related AEs.
Increased dosing frequency: AOC-1001 increasing dosing from every 13 weeks to every 8 weeks may raise drug exposure and toxicity risk.
Unproven modality in this indication: siRNA has not yet been validated in neuromuscular diseases, whereas a few ASO therapies have received FDA approval. It remains unclear whether AOC-1001’s siRNA-based mechanism can scale safely in larger patient populations.

main sources:

Comments